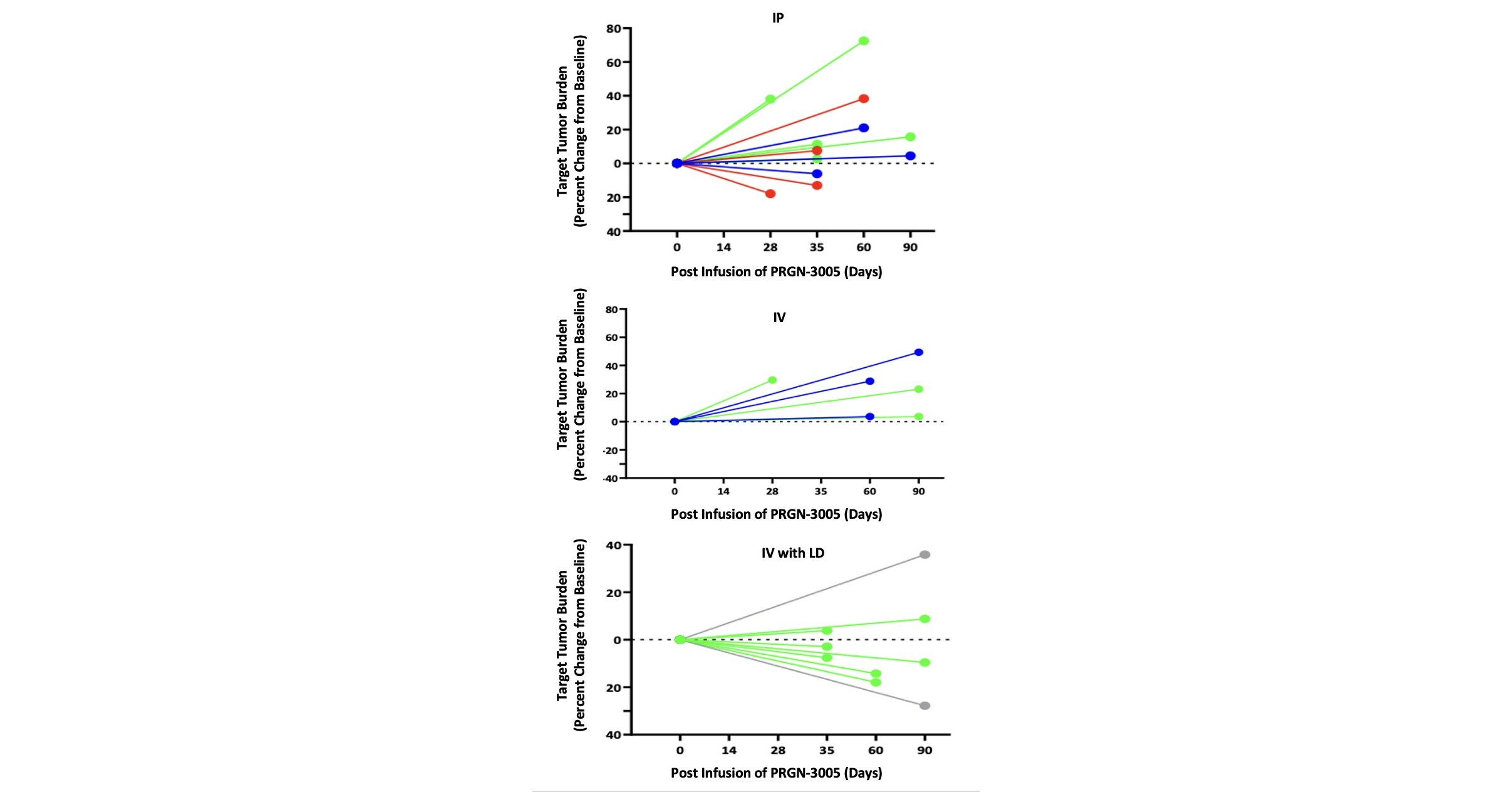
Precigen Announces Positive Phase 1 Data for PRGN-3005 Autologous UltraCAR-T® Cells Manufactured Overnight for Infusion Next Day to Advanced Stage Platinum Resistant Ovarian Cancer Patients
“We are pleased with the results of the Phase 1 study which demonstrate a favorable safety profile for PRGN-3005 UltraCAR-T. Our UltraCAR-T therapies continue to be well-tolerated with no dose limiting toxicities across our clinical stage UltraCAR-T portfolio,” said Helen Sabzevari, PhD, President and CEO of Precigen. “At this early stage, we are encouraged that … Read more